HOMER - Healthcare Orientated Machine-LEarning Research
Lcn2 in Kidney Transplantation | Sub-project of CRC Renoprotection (SFB 1365)
Therapeutic Potential of Lcn2 in Kidney Transplantation
Lipocalin-2 (Lcn2), a member of the lipocalin family, is distinctly upregulated in kidney transplants, and its level in the body fluids serves as a promising early marker of AKI, DGF and acute rejection of the renal allografts. Apart from its role as a biomarker, Lcn2 actively protects against AKI, such as IRI. However, which route the protein takes, and the functional role and mechanisms underlying Lcn2 up-regulation within the transplanted organs, remain largely unknown. We recently employed a murine model of kidney transplantation to address a potential therapeutic effect of Lcn2 to kidney allografts (Ashraf et al. Am J Transplant, 2016). Using Lcn2 deficient (Lcn2-/-) mice in both donor and recipient combinations, we demonstrated that endogenously expressed Lcn2 had little or no effect early after kidney transplantation Figure-1. However, peri-operative administration of rLcn2:Siderophore:Fe complex (rLcn2) to the recipient resulted in functional and morphological amelioration of the allograft injury.
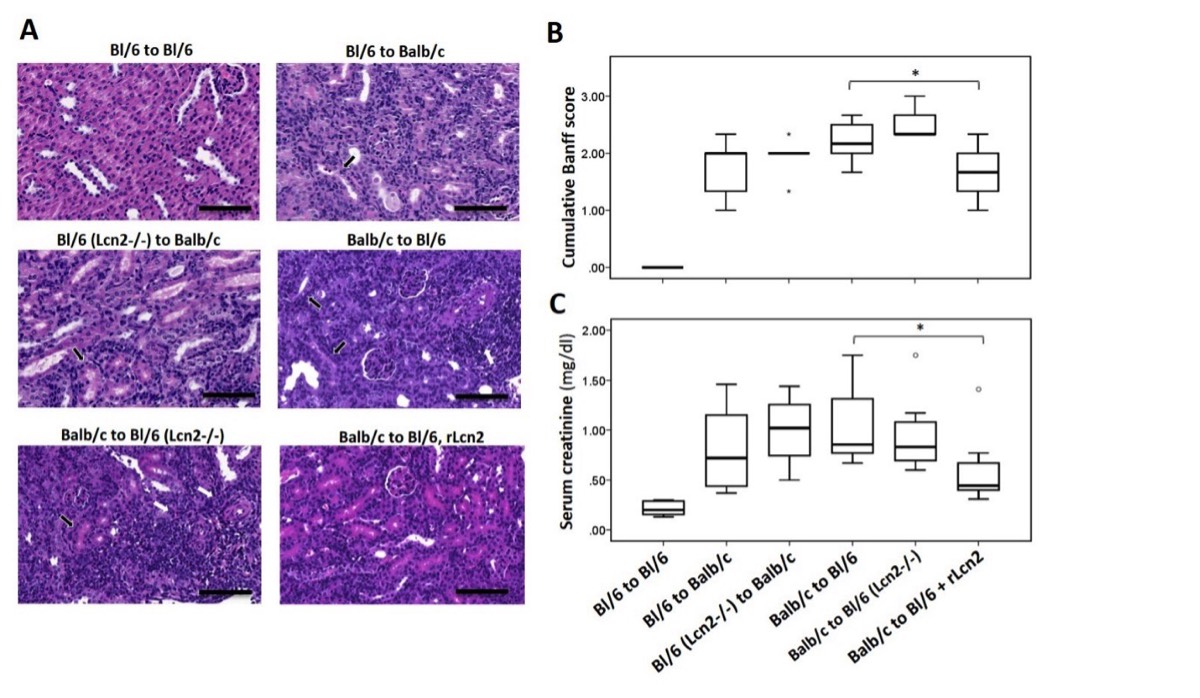
Nevertheless, the molecular and cellular events underlying the renoprotective effects of rLcn2 in kidney allografts remain unclear, which is mandatory for comprehensive translation of the therapy into clinical practice. Elucidating these events forms the main aim and primary focus of this DFG funded project. Moreover, it is essential to determine whether the Lcn2 produced in the allograft or detected in the body fluids following kidney transplantation is complexed with mammalian siderophore/Fe and may provide renoprotection to the allograft in the long run.
Therefore, the major aims of the project are:
- understanding the route and mechanisms of immunoregulation and/or cytoprotection, mediated by exogenously administered rLcn2 during the allograft damage and
- characterizing the source and nature of endogenous Lcn2 i.e. whether it is complexed with mammalian iron binding catechols and may contribute to allograft survival in the long run
Our ultimate goal is to pave the way for transplant renoprotection via rLcn2.
This is a sub project (C2) of CRC-Renoprotection (1365). The CRC is an interdisciplinary research consortium aiming at identify and targeting common pathways to kidney failure and thereby devise new treatment approaches (Spokesperson: Prof. Dr. Pontus B. Persson, Charité - Universitätsmedizin Berlin).
For more details, kindly follow the link given below:
https://nephroprotektion.charite.de/en/
Our Team

Sadia Sarfraz
Researcher



