Diffusion-weighted magnetic resonance imaging using a preclinical 1 T PET/MRI

Authors are J. Albrecht, D. Polenz, A.A. Kühl, J.M.M. Rogasch, A. Leder, I.M. Sauer, M. Babos, G. Mócsai, N. Beindorff, I.G. Steffen, W. Brenner, and E.J. Koziolek.
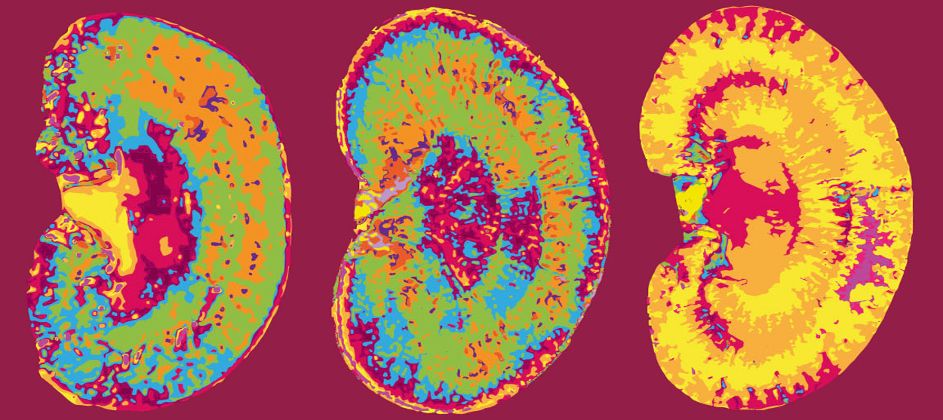
Hybrid positron emission tomography and magnetic resonance imaging (PET/MRI) scanners are increasingly used for both clinical and preclinical imaging. Especially functional MRI sequences such as diffusion-weighted imaging (DWI) are of great interest as they provide information on a molecular level, thus, can be used as surrogate biomarkers. Due to technical restrictions, MR sequences need to be adapted for each system to perform reliable imaging. There is, to our knowledge, no suitable DWI protocol for 1 Tesla PET/MRI scanners.
We established a respiratory-gated DWI protocol for a preclinical 1 T PET/MRI scanner allowing to monitor growth-related changes in ADC values of orthotopic HCC liver tumors. By monitoring the changes in tumor ADCs over time, different cellular stages were described. However, each study needs to adapt the protocol further according to their question to generate best possible results.