Engineering an endothelialized, endocrine NeoPancreas

Islet-based recellularization of decellularized, repurposed rat livers may form a transplantable Neo-Pancreas. The aim of this study is the establishment of the necessary protocols, the evaluation of the organ structure and the analysis of the islet functionality ex vivo.
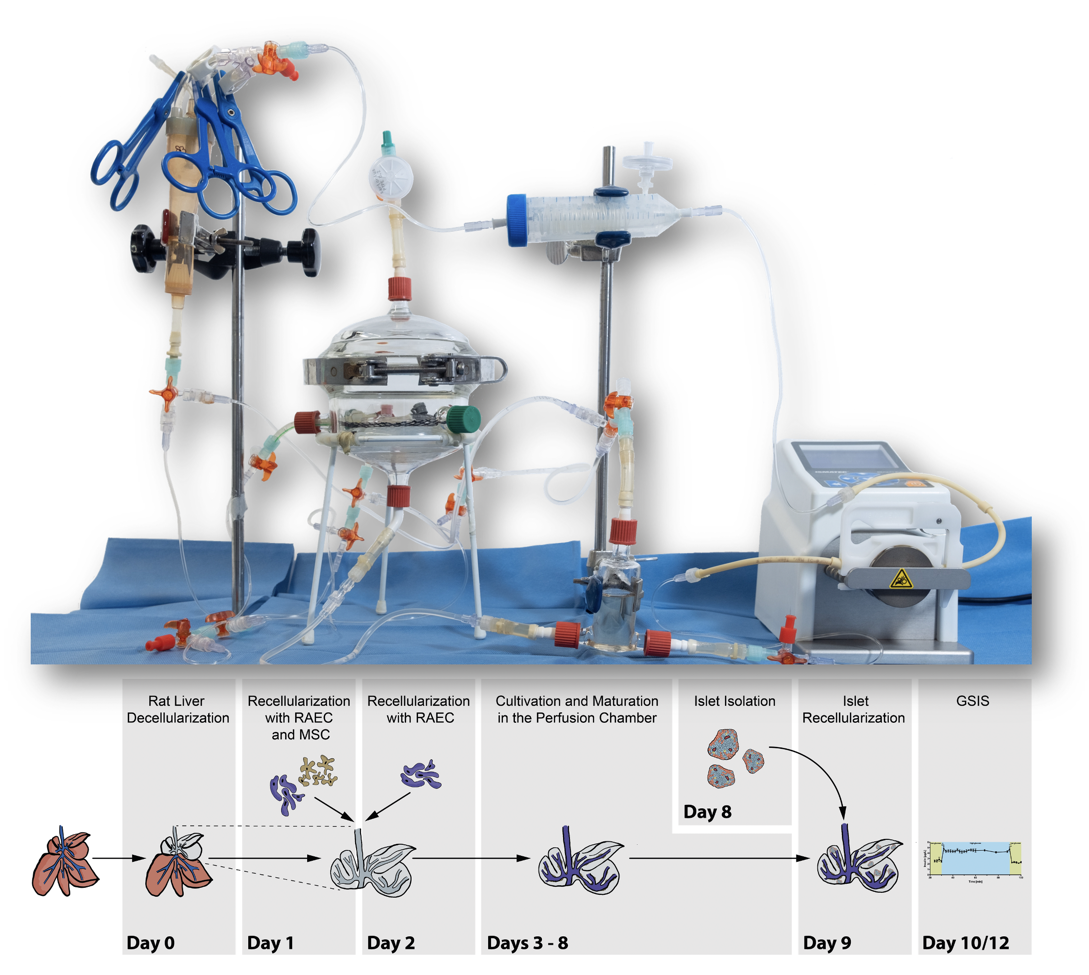
After perfusion-based decellularization of rat livers, matrices were repopulated with endothelial cells and mesenchymal stromal cells, incubated for 8 days in a perfusion chamber and finally repopulated on day 9 with intact rodent islets. Integrity and quality of re-endothelialization was assessed by histology and FITC-dextran perfusion assay. Functionality of the islets of Langerhans was determined on day 10 and day 12 via glucose stimulated insulin secretion.
Blood gas analysis variables confirmed the stability of the perfusion cultivation. Histological staining showed that cells formed a monolayer inside the intact vascular structure. These findings were confirmed by electron microscopy. Islets infused via the bile duct could histologically be found in the parenchymal space. Adequate insulin secretion after glucose stimulation after 1-day and 3-day cultivation verified islet viability and functionality after the repopulation process.
We provide the first proof-of-concept for the functionality of islets of Langerhans engrafted in a decellularized rat liver. Furthermore, a re-endothelialization step was implemented to provide implantability. This technique can serve as a bioengineered platform to generate implantable and functional endocrine Neo-Pancreases.
Authors are Hannah Everwien, Eriselda Keshi, Karl H. Hillebrandt, Barbara Ludwig, Marie Weinhart, Peter Tang, Anika S. Beierle, Hendrik Napierala, Joseph MGV Gassner, Nicolai Seiffert, Simon Moosburner, Dominik Geisel, Anja Reutzel-Selke, Benjamin Strücker, Johann Pratschke, Nils Haep, and Igor M. Sauer.